Whitley Workstation customer, BiomeBank, receives TGA (Therapeutic Goods Administration) approval for a donor derived microbiome drug. The drug - BIOMICTRA - will be used for restoration of gut microbiota in the treatment of recurrent Clostridioides difficile infection. A recent article in the publication Microbiome Times tell the story:
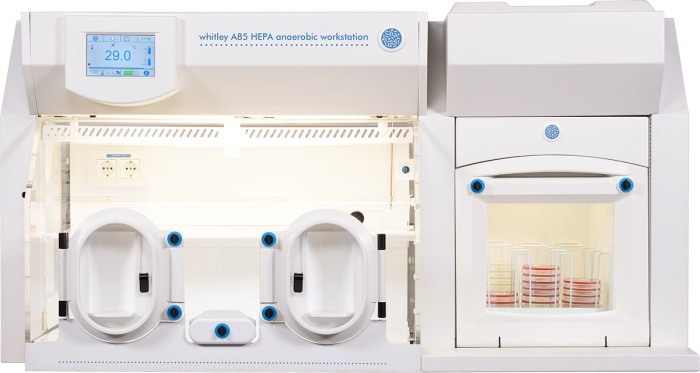
BiomeBank, based in Adelaide, Australia, are the proud owners of a Whitley A85 HEPA Workstation, which will be joined by an A55 Workstation early in 2023.


 de
de


 England
England