Faecalibacterium prausnitzii is a Gram-negative, non-spore forming, non-motile bacterium that is one of the most abundant species in the gut microbiota with approximately 5-15% detected in stool samples [1]. Its prevalence has been confirmed by 16S rRNA analyses of bacterial diversity in human faeces and colon sample, independent of cultivation. However, the percentage of F. prausnitzii detected varies between individuals depending on different factors, including whether the individual has a metabolic or intestinal disease [1]. F. prausnitzii is a very strict anaerobe, being extremely sensitive to oxygen, and was initially considered difficult to isolate and culture [1]. This issue is not exclusive to F. prausnitzii, as the gut microbiota contains many strict anaerobes [2].
Reliable growth of F. prausnitzii in pure culture is dependent on both low oxygen levels and an optimal culture medium formulation [2]. It cannot be isolated on standard anaerobic blood agar formulations such as those used in clinical microbiology laboratories.
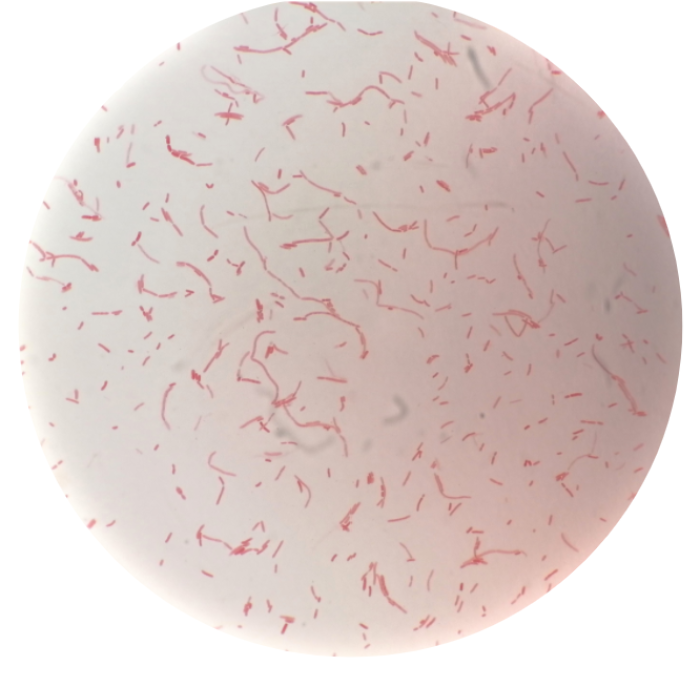
Although F. prausnitzii is a Gram-negative species, (the photograph shows a typical Gram-negative staining F. prausnitzii culture from our own laboratory) and was originally classified within the genus Fusobacterium, it has been reported as staining Gram-positive [1]. This staining behaviour is due to a lack of lipopolysaccharides in the outer membrane, so the structure of F. prausnitzii resembles Gram-positive bacteria more than Gram-negative bacteria. Indeed, 16S rRNA analyses indicate relatedness with Gram-positive bacteria of the Clostridium cluster IV, a bacterial group that efficiently ferments the plant polysaccharides found in dietary fibre.
Research into the many potential roles of F. prausnitzii has grown enormously during the last decade, initially due to an association with intestinal diseases, such as inflammatory bowel diseases (IBD) [1]. In the intestinal environment, F. prausnitzii produces the short chain fatty acid butyrate, which has a beneficial effect on colonocytes by increasing immune system maintenance and reducing inflammation [1]. Levels of this bacterium correlate with IBD: lower levels are seen in Crohn’s patients in comparison with healthy patients [1]. It may also be a useful biomarker for ulcerative colitis, specifically in the context of the Faecalibacterium – Escherichia coli ratio [8]. More recently, F. prausnitzii has been found to have a therapeutic effect in depression and anxiety (in rat studies) and may therefore have a role as a “psychobiotic” [9]. The realization that the microbiota-gut-brain axis plays a critical role in health and disease is rapidly advancing.
Microbiologists from our in-house laboratory have performed culture, enumeration and antibiotic susceptibility testing of various F. prausnitzii strains behalf of clients and for in-house research. Some of the challenges associated with culturing this species are well documented in the literature [4,5,6]. 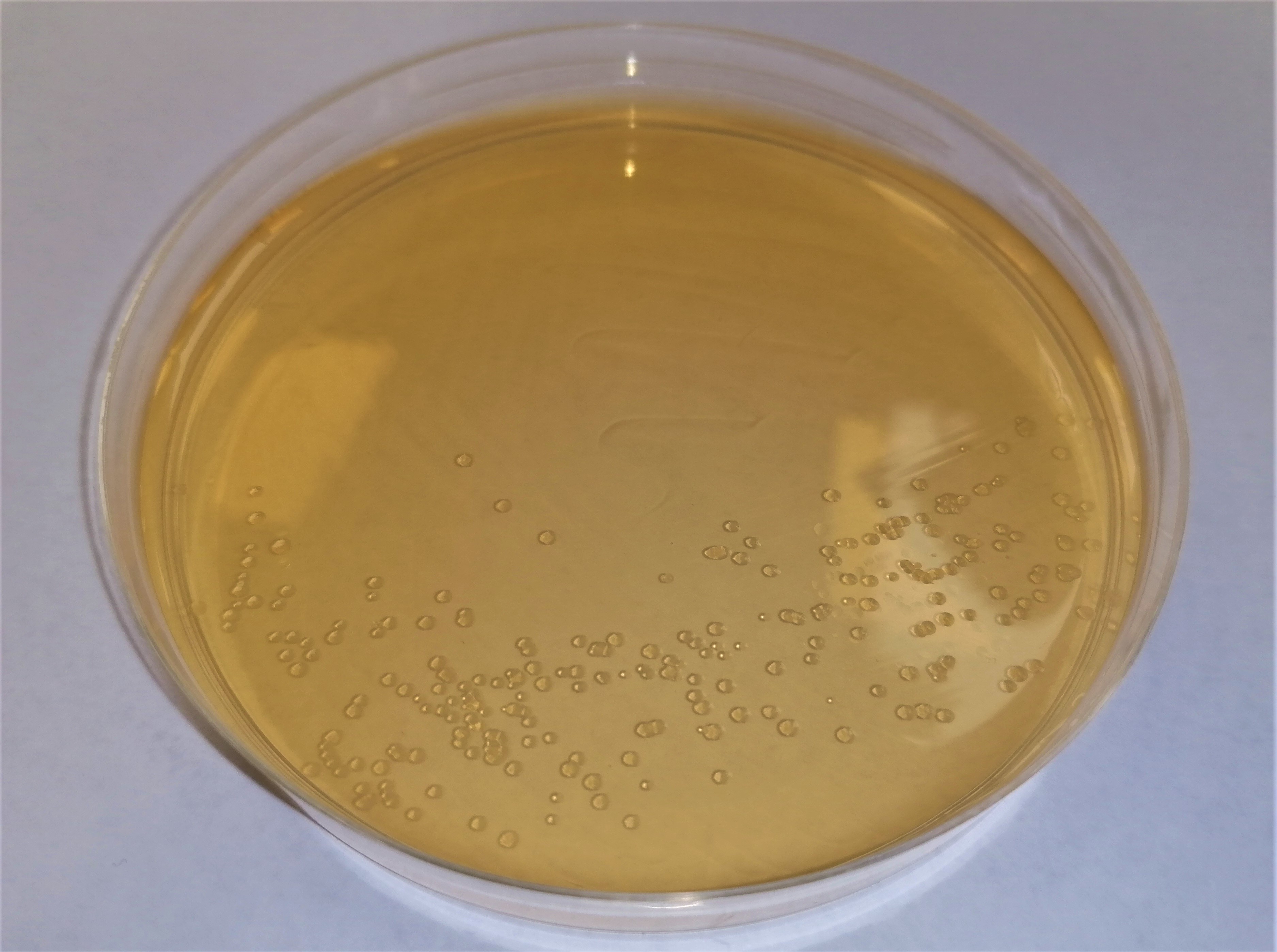 Standard media such as Fastidious Anaerobe Agar and Brucella Blood Agar tend to yield insufficient growth because F. prausnitzii requires specific fatty acids and carbohydrates [5]. In our experience, YCFA medium or the less complex YBHI medium [7] achieve reliable results. In addition to the use of an appropriate culture medium, it is essential to maintain a stringently anaerobic environment when working with this organism. Conditioning (pre-reduction) of agar plates or liquid medium within an anaerobic chamber for at least 24 hours prior to inoculation is imperative. The recommended gas mixture for Whitley Anaerobic Workstations (80% N2, 10% CO2 and 10% H2) promotes development of readily visible colonies on solid media when incubated at 35oC for 48 - 72 hours.
Standard media such as Fastidious Anaerobe Agar and Brucella Blood Agar tend to yield insufficient growth because F. prausnitzii requires specific fatty acids and carbohydrates [5]. In our experience, YCFA medium or the less complex YBHI medium [7] achieve reliable results. In addition to the use of an appropriate culture medium, it is essential to maintain a stringently anaerobic environment when working with this organism. Conditioning (pre-reduction) of agar plates or liquid medium within an anaerobic chamber for at least 24 hours prior to inoculation is imperative. The recommended gas mixture for Whitley Anaerobic Workstations (80% N2, 10% CO2 and 10% H2) promotes development of readily visible colonies on solid media when incubated at 35oC for 48 - 72 hours.
References
- Miquel S, Martín R, Rossi O, Bermúdez-Humarán L, Chatel J, Sokol H et al. (2013( Faecalibacterium prausnitzii and human intestinal health. Current Opinion in Microbiology 16, 255-261.
- Browne H, Forster S, Anonye B, Kumar N, Neville B, Stares M et al. (2016) Culturing of ‘unculturable’ human microbiota reveals novel taxa and extensive sporulation. Nature 533, 543-546.
- Fitzgerald C, Shkoporov A, Sutton T, Chaplin A, Velayudhan V, Ross R et al. (2018) Comparative analysis of Faecalibacterium prausnitzii genomes shows a high level of genome plasticity and warrants separation into new species-level taxa. BMC Genomics. 19, 931.
- Martín R, Miquel S, Benevides L, Bridonneau C, Robert V, Hudault S, et al (2017) Functional Characterization of Novel Faecalibacterium prausnitzii Strains Isolated from Healthy Volunteers: A Step Forward in the Use of F. prausnitzii as a Next-Generation Probiotic. Frontiers in Microbiology 8, 1226
- Duncan SH, Hold GL, Harmsen HJM, Stewart CS, Flint HJ (2002) Growth requirements and fermentation products of Fusobacterium prausnitzii, and a proposal to reclassify it as Faecalibacterium prausnitzii gen. nov., comb. nov. Int J Syst Evol Microbiol 52, 2141-2146. https://doi.org/10.1099/00207713-52-6-2141
- Foditsch C, Santos TM, Teixeira AG, Pereira RV, Dias JM, Gaeta N, Bicalho RC (2014) Isolation and characterization of Faecalibacterium prausnitzii from calves and piglets. PLoS One 9(12):e116465. https://doi.org/10.1371/journal.pone.0116465
- Wrzosek L, Miquel S, Noordine ML, Bouet S, Joncquel Chevalier-Curt M, Robert V, Philippe C, Bridonneau C, Cherbuy C, Robbe-Masselot C, Langella P, Thomas M (2013) Bacteroides thetaiotaomicron and Faecalibacterium prausnitzii influence the production of mucus glycans and the development of goblet cells in the colonic epithelium of a gnotobiotic model rodent. BMC Biol. 11:61. https://doi.org/10.1186/1741-7007-11-61
- Lopez-Siles, M., Duncan, S., Garcia-Gil, L. et al.(2017) Faecalibacterium prausnitzii: from microbiology to diagnostics and prognostics. ISME J 11, 841–852. https://doi.org/10.1038/ismej.2016.176
- Hao Z, Wang W, Guo R, Liu H. (2019) Faecalibacterium prausnitzii (ATCC 27766) has preventive and therapeutic effects on chronic unpredictable mild stress-induced depression-like and anxiety-like behavior in rats. Psychoneuroendocrinology. 104, 132-142. https://doi.org/10.1016/j.psyneuen.2019.02.025


 de
de


 England
England